Birth control pills recalled due to packaging mistake
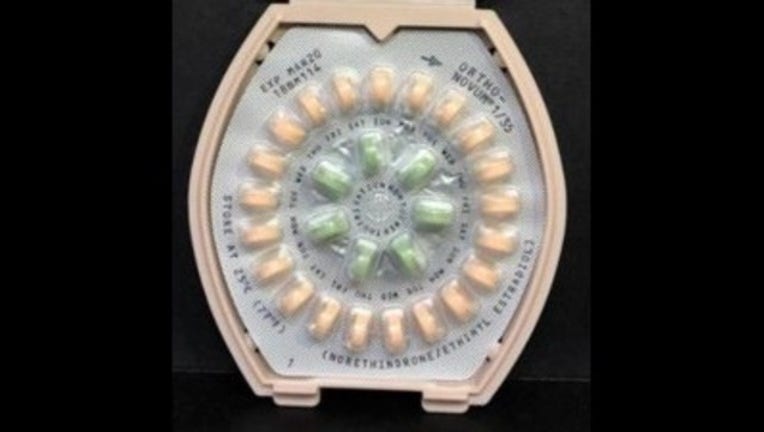
Photo credit U.S. Food & Drug Administration
(KMSP) - A pharmaceutical company has issued a voluntary recall on a birth control product, citing that the patient information provided inside the affected packages does not include the appropriate instructions for the dispenser.
According to the release, Janssen Pharmaceuticals, Inc. has initiated a voluntary recall of one lot of ORTHO-NOVUM 1/35 (norethindrone/ethinyl estradiol) Tablets and two lots of ORTHO-NOVUM 7/7/7 (norethindrone/ethinyl estradiol) Tablets to the pharmacy level.
The potential risk of taking the tablets without the appropriate instruction is that the consumer could take the pills in the incorrect order (still receiving an effective dose) or could take an inactive "reminder" pill instead of an "active" pill which could lead to breakthrough bleeding or an unintended pregnancy.
For more information including the list of affected lots, click here.

