Epidemiologists urge creation of ‘smart testing’ system in US to minimize climbing COVID-19 deaths
LOS ANGELES - A report published on May 20 by the University of Minnesota’s Center for Infectious Disease Research and Policy (CIDRAP) is calling for greater coordination by state and federal agencies in developing a more accessible and functional coronavirus testing infrastructure across the U.S.
RELATED: CoronavirusNOW.com, FOX launches national hub for COVID-19 news and updates
Citing an “unprecedented” importance and to meet testing demands, the report says that more needs to be done at the federal level to ensure those demands are met.
According to the report, a reliance on public health and commercial laboratories has limited critical potential for coronavirus testing. It urges the need for more support between state and local institutions to finance hospital-based SARSCoV-2 testing
“Current plans for clinical and public health laboratory testing do not sufficiently address the infrastructure needed to perform such tests. Critical guidance and coordination at the federal level is needed to meet the SARS-CoV-2 testing demand,” the report says.
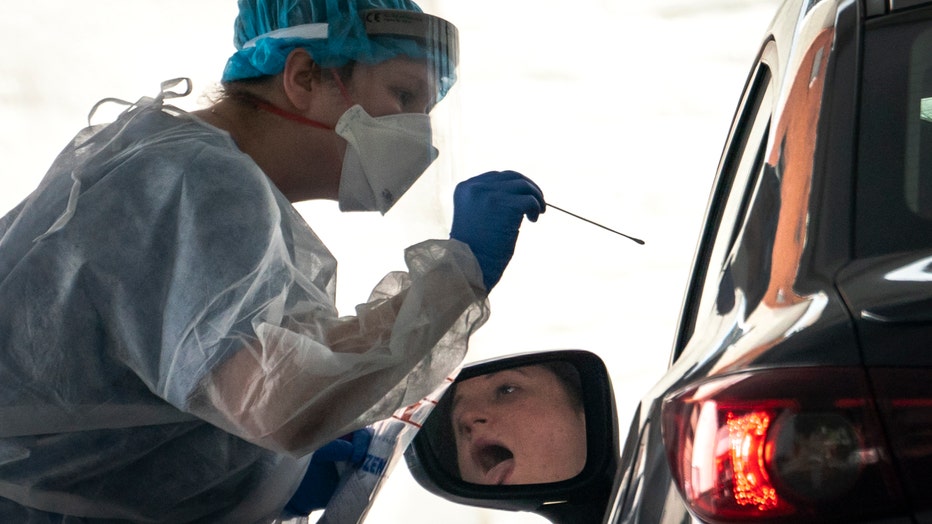
FILE - A medical professional administers a coronavirus test at a drive-thru testing site run by George Washington University Hospital, May 26, 2020 in Washington, DC. (Photo by Drew Angerer/Getty Images)
Report authors added that expanding COVID-19 testing is critical to combating the disease.
RELATED: ‘It is affecting every age group’: 3D video shows extensive damage to lungs caused by COVID-19
"For testing to be maximally effective, coordination across the system and across jurisdictions is necessary. Ideally, this requires federal guidance, leadership and support, with strong jurisdictional buy-in at the state and local levels."
The report also calls on the U.S. secretary of Health and Human Services (HHS) to appoint a panel of national experts to ensure testing demands are met, adding that every state should develop a comprehensive testing plan to ensure timely results in an attempt to minimize transmission.
RELATED: Asymptomatic individuals may be rapidly spreading COVID-19, according to researchers
Antibody testing has expanded in the past month as a possible means at understanding just how many Americans have been infected by the new coronavirus. But the report warns that while these tests can provide evidence of current or previous infection, they are not intended “as a diagnostic tool for confirming infection.”
RELATED: How should I clean and store my face mask?
“Nor is it clear if having antibodies to the virus protects someone from being infected again in the future,” the report says.
“The panel should include representatives from public health, clinical laboratory, and medicine; the laboratory testing research and development, marketing, and product support industries; ethicists; legal scholars; and elected officials,” it says.
While noting that the need for testing will most likely and in most cases already have outpaced testing capacity, CIDRAP says it recommends priority testing based on the following hierarchy:
- Those with symptoms who are critically ill and hospitalized
- Symptomatic healthcare workers and first responders, symptomatic individuals in congregate living facilities, and symptomatic essential workers
- Symptomatic individuals in the community
- Asymptomatic people living in congregate settings (e.g., long-term care facilities or homeless shelters) for the purpose of infection control



